- Blog
- Black ish season 2 free torrent download
- Sins of a solar empire rebellion trainers
- Skyrim all alchemy recipes mod
- Bias amp 2 model listy
- Fortnite nsfw discord server
- Periodic table and valence electrons
- Seven habits of highly effective people chapters
- Activ life led bike wheel lights installation
- New jay z beyonce video
- Making the toy story 3 incinerator
- Windows keyboard shortcuts
- Amy frosty freeze
- Black bear diner menu pictures
- Bingo caller machine 1 75
- Videolan movie maker free download
- Bluej example projects download
- Mpps v18 main tricore multiboot
- Wurst hacked client minecraft bedrock
- Program chamberlain garage door keypad
- Adobe illustrator chinese font free download
- Linkedin resume template 2020
- Modern script fonts free
- Sex mod sims 4 free download
- Weight loss tracker template printable
- The ultimate slide position chart for trombones
- Easeus partition master license code 2020 free
- Wii play tanks 2 players
- Among us dead body coloring pages
- Battletech record sheets succession wars records
- Freeware sound booster windows 10 completely free
- 2d rocket league mobile
- Best rust aimbot
- Camtasia 9 free video assets
- Minecraft steve dabbing
- Budget for business plan template
- Sims 4 stripper career mod download
- International commercial invoice template excel
- Apartment inspection checklist move out
- Funeral home pleasant unity pa
- Sight words worksheets for esl students
- Dxo photolab presets free
- Among us cool logo
- Resume templates 2019
- Ditching bucket for mini ihi excavator
- Sonic mania game play
- Concrete half block near me
- Patriots vs raiders free live stream

To achieve the noble gas configuration, alkali metals need to lose one electron (valence is one), whereas alkaline earth metals need to remove two. Elements in the same group share similar characteristics, like reactivity. However, as you go down the periodic table, there are more filled shells, so the valence electron are further away from the nucleus, and so are not as tightly held in, increasing the atomic radius. The periodic table organizes elements into groups and periods based on their chemical and physical properties. For example, a quick glance at Figure AT5.2. In the same group, the effective nuclear charge does not change. That way, we can get a better look at the relationship. Often it is useful to plot data on a graph. As a result, fluorine is usually thought of as the most electronegative element. The electron configuration of a oxygen atom is. While core electrons are not involved in bonding, they influence the chemical reactivity of an atom. However, on many scales, fluorine would be the most electronegative atom here. Generally, valence electrons can participate in the formation of chemical bonding, but core electrons cannot.
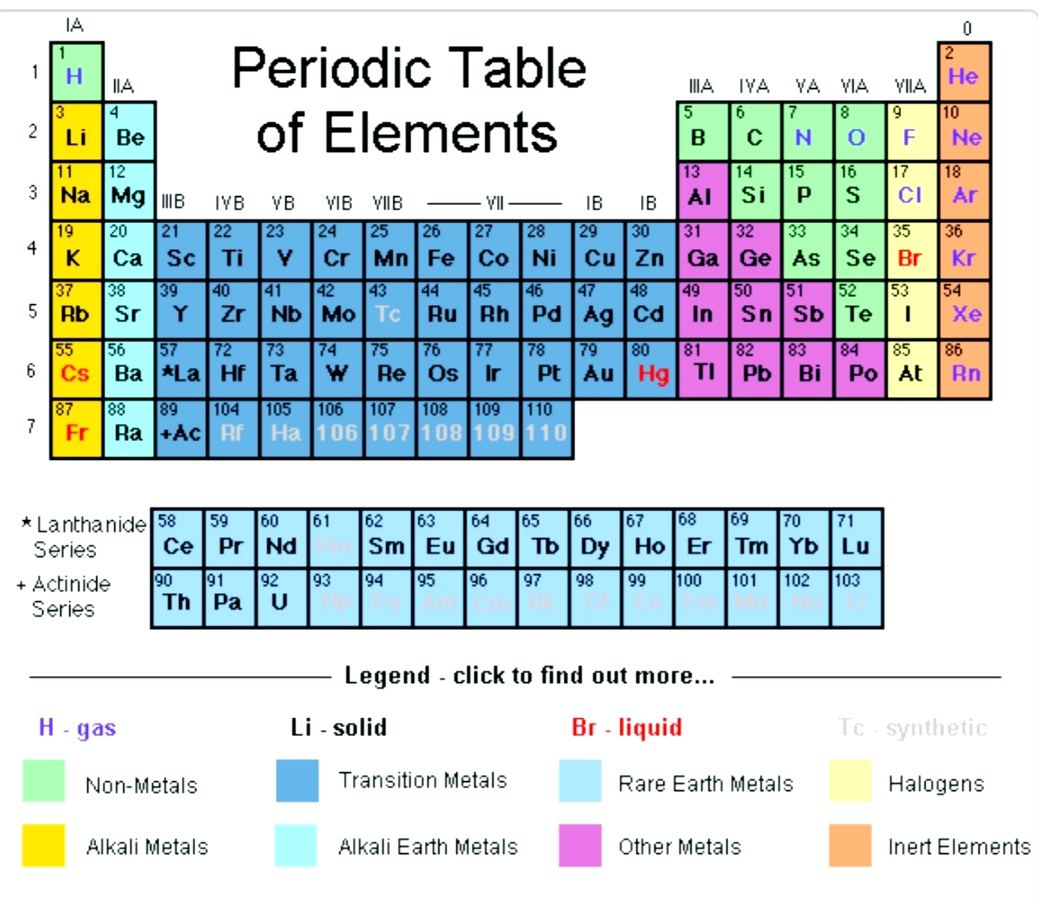
As a result, noble gases are also given electronegativity values on this scale.

The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. \): The Allen electronegativity values of the second-row elements.
